SmartSEC™ HT EV Isolation System for Serum & Plasma
- Powerful—Accelerate EV biomarker, diagnostics, and therapeutics R&D with high-throughput, 96-well plate-based EV isolation
- Fast—Isolate EVs from as many as 96 samples in less than one hour
- Flexible—Use all the wells in a single run or run a partial plate and save wells for later use
- Easy to use—Load serum or plasma directly onto the SmartSEC HT plate after a brief centrifugation—no need to pre-treat plasma!
- High-performance—Achieve levels of purity and yield better than ultracentrifugation
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| SSEC096A-1 | SmartSEC™ HT EV Isolation System (for Serum & Plasma) | 96 Reactions | $3311 |
|
||||
Overview
Overview
Overcome throughput barriers in extracellular vesicle research with SmartSEC™ HT
While extracellular vesicles (EVs) hold a lot of promise for diagnostic and therapeutic use, research and development has been hampered by the lack of a robust, high-throughput isolation method that would facilitate simultaneous collection/comparison of EVs from multiple sources. To overcome this challenge SBI developed the SmartSEC™ HT EV Isolation System for Serum & Plasma, the first kit on the market that enables EV isolation in a 96-well plate-based format. With SmartSEC HT, you get high yields of highly pure EVs with the high-throughputs needed for biomarker discovery, diagnostic development, therapeutic development, and more.
The SmartSEC HT EV Isolation System is a proprietary chromatography-based EV isolation method that comes in a 96-well plate format*. SmartSEC HT combines all the benefits of size exclusion chromatography (SEC)—purity, yield, reproducibility, and preservation of EV integrity—with a contaminant trapping feature that overcomes the limitations of conventional SEC for a fast, easy, and high-throughput EV isolation workflow.
- Powerful—Accelerate EV biomarker, diagnostics, and therapeutics R&D with high-throughput, 96-well plate-based EV isolation
- Fast—Isolate EVs from as many as 96 samples in less than one hour
- Flexible—Use all the wells in a single run or run a partial plate and save wells for later use
- Easy to use—Load serum or plasma directly onto the SmartSEC HT plate after a brief centrifugation—no need to pre-treat plasma!
- High-performance—Achieve levels of purity and yield better than ultracentrifugation
- Versatile—Isolated EVs are compatible with most downstream applications
Each SmartSEC HT kit comes with optimized amounts of SmartSEC resin already aliquoted into a 96-well filter plate, SmartSEC Isolation Buffer, and 2 collection plates. Each well of the filter plate can be loaded with 250 – 500 µL of serum or plasma and, if desired, unused wells can be preserved for future use. The entire SmartSEC HT System is compatible with standard manual and automated liquid handling systems.
If you're interested in a trial size of our SmartSEC HT technology, please contact tech@systembio.com
*For processing individual samples we recommend using SmartSEC Single, which delivers similar purity and yields as SmartSEC™ HT but in a single reaction format.References
How It Works
How It Works
Isolate EVs from up to 96 samples in under an hour
The SmartSEC HT workflow is fast, easy, and high-throughput (Figure 1). Simply prep the filter plate, apply 250 – 500 µL of cleared serum or plasma directly to each well, incubate, and centrifuge to elute the first fraction. Add an equal volume of SmartSEC Isolation Buffer and centrifuge again into a clean plate to elute the second fraction. Depending on the volume of sample loaded, the majority of EVs will be collected in either the first or second elution (we recommend collecting the two fractions separately for analysis and choosing to pool or not depending on your needs).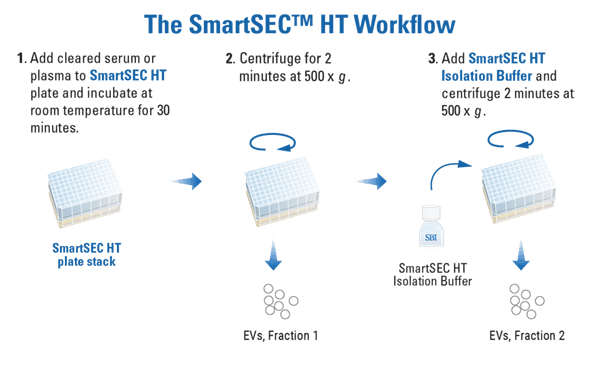
Video 1. Using SmartSEC HT.
Video 2. Preserving wells for later use.
Supporting Data
Supporting Data
See the purity and yields achievable with SmartSEC HT
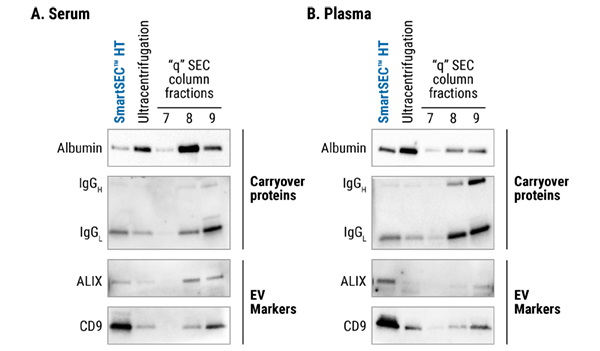
Figure 1. SmartSEC™ HT delivers high yields of EVs with low amounts of undesirable carry-over protein. EVs were prepared from 500 µL of serum (A) or plasma (B) using the indicated methods. For Western blot analysis, 1 µg protein equivalent from the first fraction was loaded into each lane. SmartSEC™ HT performs better than ultracentrifugation and a competitor’s “q” SEC column.
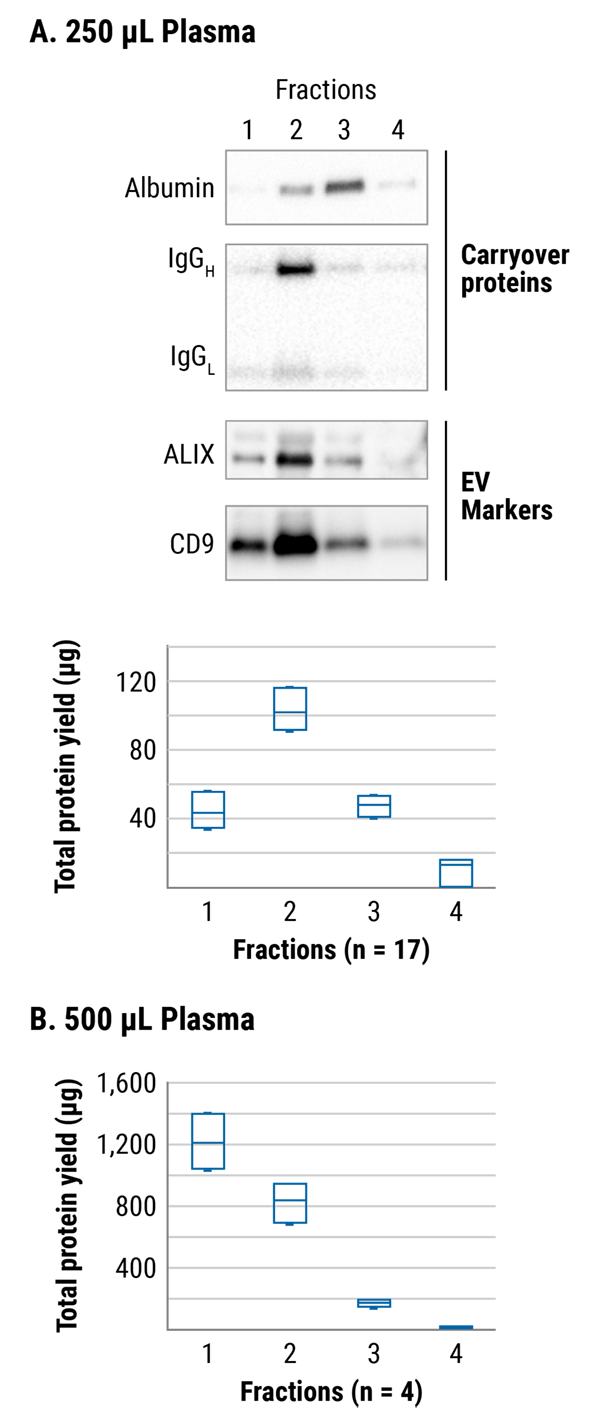
Figure 2. The majority of EVs elute in the first two fractions. As is typical for SEC, the elution profile is dependent on the input sample and eluent volumes. For 250 µL of input plasma (A), fraction 2 contains the most EVs whereas for 500 µL of input plasma (B), fraction 1 contains the most EVs. When first using SmartSEC HT, we recommend isolating fractions 1 and 2 separately, analyzing, and then choosing to pool or not depending on your needs.
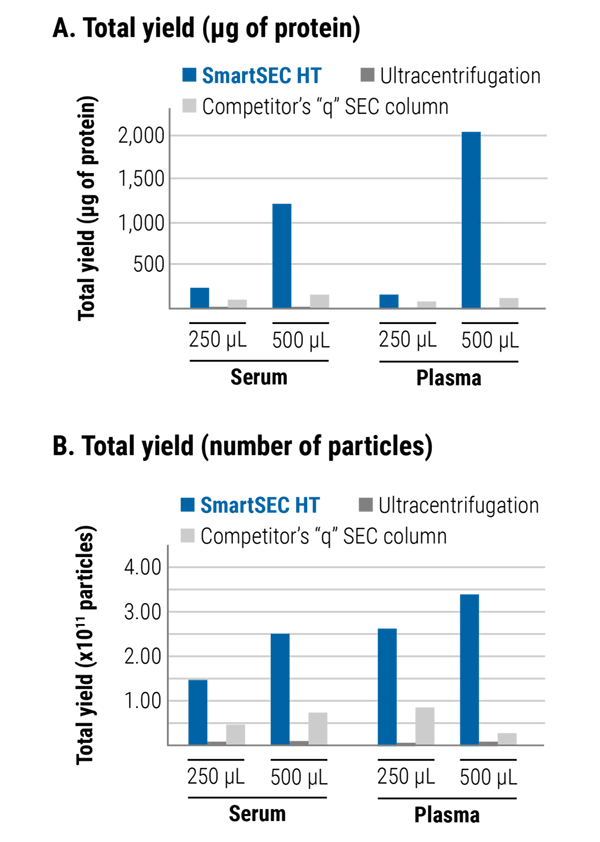
Figure 3. SmartSEC™ HT delivers higher yields than ultracentrifugation and a competitor’s “q” SEC column. We prepared EVs from 250 µL and 500 µL of serum and plasma using the indicated methods and determined yield using using (A) a Qubit protein assay and (B) fluorescent nanoparticle tracking analysis (fNTA). Table 1 provides a tabular summary of the plotted data values. By both EV yield methods, SmartSEC HT outperforms the other methods.
| Sample | Sample Volume | EV Isolation Method | EV Yield (total protein)* | EV Yield (number of particles)** |
|---|---|---|---|---|
| Serum | 250 µL | SmartSEC HT | 239 µg | 1.48 x 1011 |
| Ultracentrifugation | 4.34 µg | 0.08 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 101.6 µg | 0.44 x 1011 | ||
| 500 µL | SmartSEC HT | 1,220 µg | 2.50 x 1011 | |
| Ultracentrifugation | 5.4 µg | 0.05 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 152.5 µg | 0.72 x 1011 | ||
| Plasma | 250 µL | SmartSEC HT | 159 µg | 2.62 x 1011 |
| Ultracentrifugation | 3.77 µg | 0.014 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 76 µg | 0.80 x 1011 | ||
| 500 µL | SmartSEC HT | 2,026 µg | 3.40 x 1011 | |
| Ultracentrifugation | 4.4 µg | 0.03 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 112 µg | 0.277 x 1011 | ||
| *EV yield determined using a Qubit protein assay | ||||
| **EV yield determined by fluorescent nanoparticle tracking analysis (fNTA) | ||||
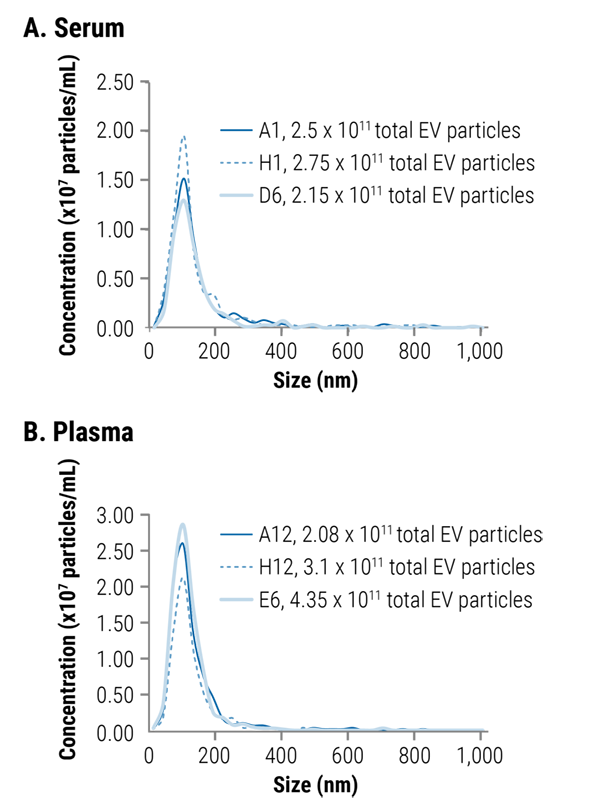
Figure 4. The SmartSEC HT plate delivers highly consistent EV isolation across the plate. To demonstrate the uniformity of performance across the SmartSEC HT plate, we loaded 500 µL of serum (A) and plasma (B) into different wells, isolated EVs and analyzed by fNTA (figure legend indicates the well position and total EV yield of each sample). For both serum and plasma samples, the total number of EV particles and the particle size distribution was highly reproducible from well-to-well.
FAQs
Documentation
Citations
Related Products
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| SSEC096A-1 | SmartSEC™ HT EV Isolation System (for Serum & Plasma) | 96 Reactions | $3311 |
|
||||
Overview
Overview
Overcome throughput barriers in extracellular vesicle research with SmartSEC™ HT
While extracellular vesicles (EVs) hold a lot of promise for diagnostic and therapeutic use, research and development has been hampered by the lack of a robust, high-throughput isolation method that would facilitate simultaneous collection/comparison of EVs from multiple sources. To overcome this challenge SBI developed the SmartSEC™ HT EV Isolation System for Serum & Plasma, the first kit on the market that enables EV isolation in a 96-well plate-based format. With SmartSEC HT, you get high yields of highly pure EVs with the high-throughputs needed for biomarker discovery, diagnostic development, therapeutic development, and more.
The SmartSEC HT EV Isolation System is a proprietary chromatography-based EV isolation method that comes in a 96-well plate format*. SmartSEC HT combines all the benefits of size exclusion chromatography (SEC)—purity, yield, reproducibility, and preservation of EV integrity—with a contaminant trapping feature that overcomes the limitations of conventional SEC for a fast, easy, and high-throughput EV isolation workflow.
- Powerful—Accelerate EV biomarker, diagnostics, and therapeutics R&D with high-throughput, 96-well plate-based EV isolation
- Fast—Isolate EVs from as many as 96 samples in less than one hour
- Flexible—Use all the wells in a single run or run a partial plate and save wells for later use
- Easy to use—Load serum or plasma directly onto the SmartSEC HT plate after a brief centrifugation—no need to pre-treat plasma!
- High-performance—Achieve levels of purity and yield better than ultracentrifugation
- Versatile—Isolated EVs are compatible with most downstream applications
Each SmartSEC HT kit comes with optimized amounts of SmartSEC resin already aliquoted into a 96-well filter plate, SmartSEC Isolation Buffer, and 2 collection plates. Each well of the filter plate can be loaded with 250 – 500 µL of serum or plasma and, if desired, unused wells can be preserved for future use. The entire SmartSEC HT System is compatible with standard manual and automated liquid handling systems.
If you're interested in a trial size of our SmartSEC HT technology, please contact tech@systembio.com
*For processing individual samples we recommend using SmartSEC Single, which delivers similar purity and yields as SmartSEC™ HT but in a single reaction format.References
How It Works
How It Works
Isolate EVs from up to 96 samples in under an hour
The SmartSEC HT workflow is fast, easy, and high-throughput (Figure 1). Simply prep the filter plate, apply 250 – 500 µL of cleared serum or plasma directly to each well, incubate, and centrifuge to elute the first fraction. Add an equal volume of SmartSEC Isolation Buffer and centrifuge again into a clean plate to elute the second fraction. Depending on the volume of sample loaded, the majority of EVs will be collected in either the first or second elution (we recommend collecting the two fractions separately for analysis and choosing to pool or not depending on your needs).
Video 1. Using SmartSEC HT.
Video 2. Preserving wells for later use.
Supporting Data
Supporting Data
See the purity and yields achievable with SmartSEC HT

Figure 1. SmartSEC™ HT delivers high yields of EVs with low amounts of undesirable carry-over protein. EVs were prepared from 500 µL of serum (A) or plasma (B) using the indicated methods. For Western blot analysis, 1 µg protein equivalent from the first fraction was loaded into each lane. SmartSEC™ HT performs better than ultracentrifugation and a competitor’s “q” SEC column.

Figure 2. The majority of EVs elute in the first two fractions. As is typical for SEC, the elution profile is dependent on the input sample and eluent volumes. For 250 µL of input plasma (A), fraction 2 contains the most EVs whereas for 500 µL of input plasma (B), fraction 1 contains the most EVs. When first using SmartSEC HT, we recommend isolating fractions 1 and 2 separately, analyzing, and then choosing to pool or not depending on your needs.

Figure 3. SmartSEC™ HT delivers higher yields than ultracentrifugation and a competitor’s “q” SEC column. We prepared EVs from 250 µL and 500 µL of serum and plasma using the indicated methods and determined yield using using (A) a Qubit protein assay and (B) fluorescent nanoparticle tracking analysis (fNTA). Table 1 provides a tabular summary of the plotted data values. By both EV yield methods, SmartSEC HT outperforms the other methods.
| Sample | Sample Volume | EV Isolation Method | EV Yield (total protein)* | EV Yield (number of particles)** |
|---|---|---|---|---|
| Serum | 250 µL | SmartSEC HT | 239 µg | 1.48 x 1011 |
| Ultracentrifugation | 4.34 µg | 0.08 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 101.6 µg | 0.44 x 1011 | ||
| 500 µL | SmartSEC HT | 1,220 µg | 2.50 x 1011 | |
| Ultracentrifugation | 5.4 µg | 0.05 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 152.5 µg | 0.72 x 1011 | ||
| Plasma | 250 µL | SmartSEC HT | 159 µg | 2.62 x 1011 |
| Ultracentrifugation | 3.77 µg | 0.014 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 76 µg | 0.80 x 1011 | ||
| 500 µL | SmartSEC HT | 2,026 µg | 3.40 x 1011 | |
| Ultracentrifugation | 4.4 µg | 0.03 x 1011 | ||
| Competitor's "q" SEC column (fractions 7, 8, 9 pooled) | 112 µg | 0.277 x 1011 | ||
| *EV yield determined using a Qubit protein assay | ||||
| **EV yield determined by fluorescent nanoparticle tracking analysis (fNTA) | ||||

Figure 4. The SmartSEC HT plate delivers highly consistent EV isolation across the plate. To demonstrate the uniformity of performance across the SmartSEC HT plate, we loaded 500 µL of serum (A) and plasma (B) into different wells, isolated EVs and analyzed by fNTA (figure legend indicates the well position and total EV yield of each sample). For both serum and plasma samples, the total number of EV particles and the particle size distribution was highly reproducible from well-to-well.

