Basic Exo-Flow Capture Kit
- Large-sized magnetic beads increase the efficiency of exosome capture
- Reversible Exo-FITC and Exo-APC stains can be completely removed after FACS
- Exosome Elution Buffer simultaneously removes Exo-FITC (or Exo-APC) and elutes exosomes from the magnetic beads for downstream applications such as functional studies
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CSFLOWBASICA-1 | Basic Exo-Flow kit without any coupled antibodies (Uncoupled magnetic streptavidin beads, Wash and Elution Buffers, Exo-FITC stain) | 100 Reactions | $1140 |
|
||||
Overview
Overview
Customize your Exo-Flow Capture Kit with your own antibody
The Basic Exo-Flow Capture Kit allows you to take advantage of our robust Exo-Flow technology with any exosome marker of your choice. Aside from the biotinylated antibody, the kit has all the same reagents as our other Exo-Flow Kits—magnetic streptavidin beads, wash and elution buffers, and reversible Exo-FITC stain (Cat.# EXOFLOW800A-1)—you supply the biotinylated antibody, FACS, and the optional magnetic stand (Cat.# EXOFLOW700A-1). Our high-quality kit components ensure reliable, reproducible exosome purification based on the exosome surface marker of your choice. And with our larger-than-typical bead size (9.1 μm diameter) exosome capture is highly efficient, assisting with recovery of rare exosome populations.
Note that we also offer reversible Exo-APC (Cat.# Exo-FLOW810A-1) as an alternative stain to Exo-FITC.- Large-sized magnetic beads increase the efficiency of exosome capture
- Reversible Exo-FITC and Exo-APC stains can be completely removed after FACS
- Exosome Elution Buffer simultaneously removes Exo-FITC (or Exo-APC) and elutes exosomes from the magnetic beads for downstream applications such as functional studies

Beyond the Basic Kit
To facilitate the widest range of studies, SBI built the Exo-Flow system to be highly modular. In addition to the Basic Exo-Flow Kit, we offer a family of kits with different, well-validated biotinylated antibodies.| Cat.# | Kit |
|---|---|
| EXOFLOW100A-1 | CD9 Exo-Flow Capture Kit |
| EXOFLOW150A-1 | Tetraspanin Exo-Flow Combo Capture Kit |
| EXOFLOW200A-1 | CD31 Exo-Flow Capture Kit |
| EXOFLOW210A-1 | CD44 Exo-Flow Capture Kit |
| EXOFLOW300A-1 | CD63 Exo-Flow Capture Kit |
| EXOFLOW400A-1 | CD81 Exo-Flow Capture Kit |
| EXOFLOW500A-1 | Rab5b Exo-Flow Capture Kit |
| EXOFLOW600A-1 | HLA-G Exo-Flow Capture Kit |
| EXOFLOW610A-1 | CD14 Exo-Flow Capture Kit |
| EXOFLOW620A-1 | CD68 Exo-Flow Capture Kit |
| EXOFLOW630A-1 | EpCAM Exo-Flow Capture Kit |
| EXOFLOW660A-1 | CD73 Exo-Flow Capture Kit |
References
How It Works
How It Works
Easily purify exosomes using FACS with the Basic Exo-Flow Capture Kit
At a glance
Simply (1) couple your biotinylated antibody to the magnetic streptavidin beads, (2) use the antibody-coupled magnetic beads to capture exosomes that have been isolated using either ExoQuick® or ultracentrifugation, (3) wash away unbound exosomes, and then (4) stain with reversible Exo-FITC (excitation and emission wavelengths of 494 nm and 518 nm, respectively).
Your sample is now ready for FACS analysis.
To use the purified exosomes after FACS, add the included Exosome Elution Buffer to simultaneously remove the Exo-FITC stain and elute intact exosomes from the beads.
Supporting Data
Supporting Data
See our Exo-FLOW Capture Kits in action
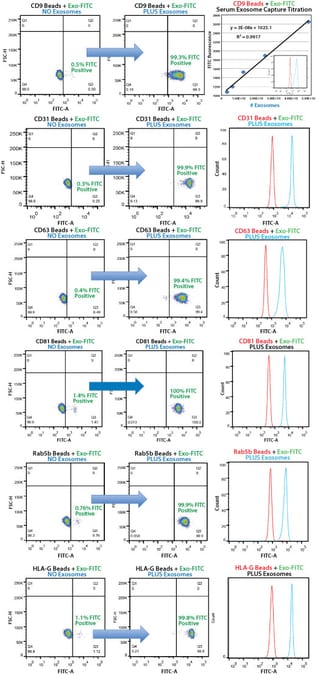
Our Exo-Flow Capture Kits deliver quantitative, highly selective exosome isolation. Bead flow separation data for exosomes secreted by HEK293 cells and captured using different Exo-Flow Capture Kits. Plots of forward scatter versus FITC intensity show that in the no exosome control, only 0.5% of particles are FITC-positive (left panels), whereas in the exosome-containing sample, 99.3% of particles are FITC-positive (middle panels). The degree of flow separation is shown in the right panels.
Human serum exosomes were isolated from 250 µL serum using ExoQuick® and the exosome pellet resuspended in 500 µL of 1x PBS. Exosome particles were added as two-fold dilutions starting at 50 µL, and then captured using the biotinylated antibody coupled to Exo-Flow beads. The FITC flow cytometric intensities are then plotted versus the number of exosome particles (determined using NanoSight).
FAQs
Documentation
Citations
Related Products
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CSFLOWBASICA-1 | Basic Exo-Flow kit without any coupled antibodies (Uncoupled magnetic streptavidin beads, Wash and Elution Buffers, Exo-FITC stain) | 100 Reactions | $1140 |
|
||||
Overview
Overview
Customize your Exo-Flow Capture Kit with your own antibody
The Basic Exo-Flow Capture Kit allows you to take advantage of our robust Exo-Flow technology with any exosome marker of your choice. Aside from the biotinylated antibody, the kit has all the same reagents as our other Exo-Flow Kits—magnetic streptavidin beads, wash and elution buffers, and reversible Exo-FITC stain (Cat.# EXOFLOW800A-1)—you supply the biotinylated antibody, FACS, and the optional magnetic stand (Cat.# EXOFLOW700A-1). Our high-quality kit components ensure reliable, reproducible exosome purification based on the exosome surface marker of your choice. And with our larger-than-typical bead size (9.1 μm diameter) exosome capture is highly efficient, assisting with recovery of rare exosome populations.
Note that we also offer reversible Exo-APC (Cat.# Exo-FLOW810A-1) as an alternative stain to Exo-FITC.- Large-sized magnetic beads increase the efficiency of exosome capture
- Reversible Exo-FITC and Exo-APC stains can be completely removed after FACS
- Exosome Elution Buffer simultaneously removes Exo-FITC (or Exo-APC) and elutes exosomes from the magnetic beads for downstream applications such as functional studies

Beyond the Basic Kit
To facilitate the widest range of studies, SBI built the Exo-Flow system to be highly modular. In addition to the Basic Exo-Flow Kit, we offer a family of kits with different, well-validated biotinylated antibodies.| Cat.# | Kit |
|---|---|
| EXOFLOW100A-1 | CD9 Exo-Flow Capture Kit |
| EXOFLOW150A-1 | Tetraspanin Exo-Flow Combo Capture Kit |
| EXOFLOW200A-1 | CD31 Exo-Flow Capture Kit |
| EXOFLOW210A-1 | CD44 Exo-Flow Capture Kit |
| EXOFLOW300A-1 | CD63 Exo-Flow Capture Kit |
| EXOFLOW400A-1 | CD81 Exo-Flow Capture Kit |
| EXOFLOW500A-1 | Rab5b Exo-Flow Capture Kit |
| EXOFLOW600A-1 | HLA-G Exo-Flow Capture Kit |
| EXOFLOW610A-1 | CD14 Exo-Flow Capture Kit |
| EXOFLOW620A-1 | CD68 Exo-Flow Capture Kit |
| EXOFLOW630A-1 | EpCAM Exo-Flow Capture Kit |
| EXOFLOW660A-1 | CD73 Exo-Flow Capture Kit |
References
How It Works
How It Works
Easily purify exosomes using FACS with the Basic Exo-Flow Capture Kit
At a glance
Simply (1) couple your biotinylated antibody to the magnetic streptavidin beads, (2) use the antibody-coupled magnetic beads to capture exosomes that have been isolated using either ExoQuick® or ultracentrifugation, (3) wash away unbound exosomes, and then (4) stain with reversible Exo-FITC (excitation and emission wavelengths of 494 nm and 518 nm, respectively).
Your sample is now ready for FACS analysis.
To use the purified exosomes after FACS, add the included Exosome Elution Buffer to simultaneously remove the Exo-FITC stain and elute intact exosomes from the beads.
Supporting Data
Supporting Data
See our Exo-FLOW Capture Kits in action
Our Exo-Flow Capture Kits deliver quantitative, highly selective exosome isolation. Bead flow separation data for exosomes secreted by HEK293 cells and captured using different Exo-Flow Capture Kits. Plots of forward scatter versus FITC intensity show that in the no exosome control, only 0.5% of particles are FITC-positive (left panels), whereas in the exosome-containing sample, 99.3% of particles are FITC-positive (middle panels). The degree of flow separation is shown in the right panels.
Human serum exosomes were isolated from 250 µL serum using ExoQuick® and the exosome pellet resuspended in 500 µL of 1x PBS. Exosome particles were added as two-fold dilutions starting at 50 µL, and then captured using the biotinylated antibody coupled to Exo-Flow beads. The FITC flow cytometric intensities are then plotted versus the number of exosome particles (determined using NanoSight).