Exosome Cyto-Tracer, pCT-CD81-GFP
- Stable lentivector-based system
- Ideal for co-localization studies
- Monitor exosome dynamics and functional studies in real time
- Label exosomes from primary cells, tumor cells, stem cells, and more
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CYTO124-PA-1 | pCT-CD81-GFP (pCMV, Exosome/Secretory, CD81 Tetraspanin Tag) | 10 µg | $830 |
|
||||
| CYTO124-VA-1 | pCT-CD81-GFP (pCMV, Exosome/Secretory, CD81 Tetraspanin Tag; pre-packaged virus) | >2x10^6 IFUs | $954 |
|
||||
Overview
Overview
Easily track exosomes with a CD81-GFP fusion Monitor exosome dynamics with the pCT-CD81-GFP (pCMV) Exosome Cyto-Tracer (also available as a CD81-RFP fusion, pCT-CD81-RFP). Provided as either lentivector or pre-packaged virus, this Cyto-Tracer fuses GFP to the tetraspanin CD81, which then gets packaged into exosomes produced by the transfected cells. Great for creating stable cell lines that produce GFP-labeled exosomes as well as labeling exosomes from primary cells, tumor cells, stem cells, and more.- Stable lentivector-based system
- Ideal for co-localization studies
- Monitor exosome dynamics and functional studies in real time
- Label exosomes from primary cells, tumor cells, stem cells, and more
| Promoter (Cat.#) | Cyto-Tracer | Target Location | Peptide Tag |
|---|---|---|---|
| CMV (CYTO100-PA-1/CYTO100-VA-1) MSCV (CYTO100-PB-1/CYTO100-VB-1) | pCT-Mem-GFP | Plasma Membrane | Neuromodulin |
| CMV (CYTO101-PA-1/CYTO101-VA-1) MSCV (CYTO101-PB-1/CYTO101-VB-1) | pCT-InnerMem-GFP | Inner Leaflet of Membrane | Farnesylation signal |
| CMV (CYTO102-PA-1/CYTO102-VA-1) MSCV (CYTO102-PB-1/CYTO102-VB-1) | pCT-Mito-GFP | Mitochondria | Cytochrome C oxidase VIII |
| CMV (CYTO103-PA-1/CYTO103-VA-1) | pCT-ER-GFP | Endoplasmic Reticulum | ER targeting consensus |
| CMV (CYTO104-PA-1/CYTO104-VA-1) | pCT-Golgi-GFP | Golgi Apparatus | Galactosyltrasferase |
| CMV (CYTO105-PA-1/CYTO105-VA-1) | pCT-Secretory-GFP | ER-Golgi Vesicles | Secretory consensus peptide |
| CMV (CYTO106-PA-1/CYTO106-VA-1) | pCT-Lyso-GFP | Lysosome | Cathepsin B |
| CMV (CYTO107-PA-1/CYTO107-VA-1) | pCT-Endo-GFP | Endosome | RhoB |
| CMV (CYTO107R-PA-1/CYTO107R-VA-1) | pCT-Endo-RFP | Endosome | RhoB |
| CMV (CYTO108-PA-1/CYTO108-VA-1) MSCV (CYTO108-PB-1/CYTO108-VB-1) | pCT-Pero-GFP | Peroxisome | Peroxisomal consensus signal |
| CMV (CYTO109-PA-1/CYTO109-VA-1) | pCT-Autophago-GFP | Autophagosome | LC3 |
| CMV (CYTO109R-PA-1/CYTO109R-VA-1) | pCT-Autophago-RFP | Autophagosome | LC3 |
| CMV (CYTO120-PA-1/CYTO120-VA-1) | pCT-CD63-GFP | Exosome/Secretory | CD63 Tetraspanin |
| CMV (CYTO120R-PA-1/CYTO120R-VA-1) | pCT-CD63-RFP | Exosome/Secretory | CD63 Tetraspanin |
| CMV (CYTO122-PA-1/CYTO122-VA-1) | pCT-CD9-GFP | Exosome/Secretory | CD9 Tetraspanin |
| CMV (CYTO123-PA-1/CYTO123-VA-1) | pCT-CD9-RFP | Exosome/Secretory | CD9 Tetraspanin |
| CMV (CYTO124-PA-1/CYTO124-VA-1) | pCT-CD81-GFP | Exosome/Secretory | CD81 Tetraspanin |
| CMV (CYTO125-PA-1/CYTO125-VA-1) | pCT-CD81-RFP | Exosome/Secretory | CD81 Tetraspanin |
| CMV (CYTO110-PA-1/CYTO110-VA-1) MSCV (CYTO110-PB-1/CYTO110-VB-1) | pCT-Actin-GFP | Cytoskeleton | β-actin |
| CMV (CYTO111-PA-1/CYTO111-VA-1) MSCV (CYTO111-PB-1/CYTO111-VB-1) | pCT-Tublin-GFP | Cytoskeleton | α-tublin |
| CMV (CYTO111R-PA-1/CYTO111R-VA-1) | pCT-Tublin-RFP | Cytoskeleton | α-tublin |
| CMV (CYTO112-PA-1/CYTO112-VA-1) | pCT-MAP4-GFP | Microtubule | Microtubule associated protein 4 (MAP4) |
| CMV (CYTO113-PA-1/CYTO113-VA-1) MSCV (CYTO113-PB-1/CYTO113-VB-1) | pCT-H2B-GFP | Nucleus | H2B |
| CMV (CYTO114-PA-1/CYTO114-VA-1) | pCT-Apoptosis-Luc | Caspase 3/7 activation | Cyclic inactive luciferase |
| CMV (CYTO115-PA-1/CYTO115-VA-1) | pCT-GFP-BAX | Cytosol to Mitochondria | BAX |
| CMV (CYTO116-PA-1/CYTO116-VA-1) | pCT- Catenin-GFP | Cytosol to Nucleus/Cell Membrane | β-Catenin activation |
| CMV (CYTO117-PA-1/CYTO117-VA-1) | pCT-CMV-PSD95-GFP | Dendrite Membranes | PSD-95 |
| CMV (CYTO118-PA-1/CYTO118-VA-1) MSCV (CYTO118-PB-1/CYTO118-VB-1) | pCT-Cyto-GFP | Cytosolic Compartment | GFP-Untagged |
| CMV (CYTO119-PA-1/CYTO119-VA-1) | pCT-Cyto-RFP | Cytosolic Compartment | RFP-Untagged |
| MSCV (CYTO121-PB-1/CYTO121-VB-1) | pCT-Cyto-GFpLuc | GFP and Luciferase Control | GFP and Luciferase (Untagged) |
References
How It Works
Supporting Data
Supporting Data
See some of our exosome Cyto-Tracers in action
The following figure and videos are from:
Garcia NA, et al. Glucose Starvation in Cardiomyocytes Enhances Exosome Secretion and Promotes Angiogenesis in Endothelial Cells. PLoS ONE. 2015. 10(9). PMCID: PMC4578916.
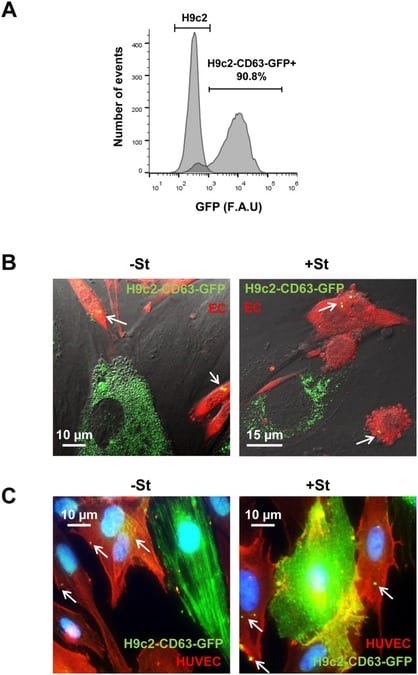
Figure 4 from Garcia, et al. Exosome transfer from CMs to ECs. (A) H9c2 transfected with pCT-CD63-GFP. FACS analysis of 90% GFP positive cells. (B) Representative images from confocal time-lapse microscopy of mouse ECs (ACTB-DsRed) (EC; red) co-cultured with H9C2-CD63-GFP cells (green), previously cultured for 24 h in +/-St medium. Exosome transfer from H9C2 CMs to EC can be observed (S1 and S2 Movies, below). White arrows show CD63-GFP structures inside ECs (C) Representative immunostaining of H9C2-CD63-GFP and HUVEC co-cultures; anti-GFP (green) and anti-CD31 (red). The images illustrate GFP fluorescence from CD63-GFP exosomes in CD31-positive cells (red) after 24 h incubation in +/-St medium. White arrows show CD63-GFP structures inside ECs.
Video S1 from Garcia, et al. Exosome transfer from H9C2-CD63-GFP (green) to endothelial DsRed cells (red) under +St conditions.
Video S2 from Garcia, et al. Exosome transfer from H9C2-CD63-GFP (green) to endothelial DsRed cells (red) under -St conditions.
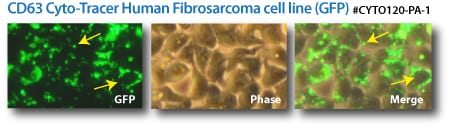
Labeled exosomes. (Top panels) CD63-GFP Cyto-Tracers transfected into a human fibrosarcoma cell line. (Bottom panels) CD9-GFP and CD9-RFP Cyto-Tracers co-transfected into HEK293 cells.
FAQs
Documentation
Citations
Related Products
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CYTO124-PA-1 | pCT-CD81-GFP (pCMV, Exosome/Secretory, CD81 Tetraspanin Tag) | 10 µg | $830 |
|
||||
| CYTO124-VA-1 | pCT-CD81-GFP (pCMV, Exosome/Secretory, CD81 Tetraspanin Tag; pre-packaged virus) | >2x10^6 IFUs | $954 |
|
||||
Overview
Overview
Easily track exosomes with a CD81-GFP fusion Monitor exosome dynamics with the pCT-CD81-GFP (pCMV) Exosome Cyto-Tracer (also available as a CD81-RFP fusion, pCT-CD81-RFP). Provided as either lentivector or pre-packaged virus, this Cyto-Tracer fuses GFP to the tetraspanin CD81, which then gets packaged into exosomes produced by the transfected cells. Great for creating stable cell lines that produce GFP-labeled exosomes as well as labeling exosomes from primary cells, tumor cells, stem cells, and more.- Stable lentivector-based system
- Ideal for co-localization studies
- Monitor exosome dynamics and functional studies in real time
- Label exosomes from primary cells, tumor cells, stem cells, and more
| Promoter (Cat.#) | Cyto-Tracer | Target Location | Peptide Tag |
|---|---|---|---|
| CMV (CYTO100-PA-1/CYTO100-VA-1) MSCV (CYTO100-PB-1/CYTO100-VB-1) | pCT-Mem-GFP | Plasma Membrane | Neuromodulin |
| CMV (CYTO101-PA-1/CYTO101-VA-1) MSCV (CYTO101-PB-1/CYTO101-VB-1) | pCT-InnerMem-GFP | Inner Leaflet of Membrane | Farnesylation signal |
| CMV (CYTO102-PA-1/CYTO102-VA-1) MSCV (CYTO102-PB-1/CYTO102-VB-1) | pCT-Mito-GFP | Mitochondria | Cytochrome C oxidase VIII |
| CMV (CYTO103-PA-1/CYTO103-VA-1) | pCT-ER-GFP | Endoplasmic Reticulum | ER targeting consensus |
| CMV (CYTO104-PA-1/CYTO104-VA-1) | pCT-Golgi-GFP | Golgi Apparatus | Galactosyltrasferase |
| CMV (CYTO105-PA-1/CYTO105-VA-1) | pCT-Secretory-GFP | ER-Golgi Vesicles | Secretory consensus peptide |
| CMV (CYTO106-PA-1/CYTO106-VA-1) | pCT-Lyso-GFP | Lysosome | Cathepsin B |
| CMV (CYTO107-PA-1/CYTO107-VA-1) | pCT-Endo-GFP | Endosome | RhoB |
| CMV (CYTO107R-PA-1/CYTO107R-VA-1) | pCT-Endo-RFP | Endosome | RhoB |
| CMV (CYTO108-PA-1/CYTO108-VA-1) MSCV (CYTO108-PB-1/CYTO108-VB-1) | pCT-Pero-GFP | Peroxisome | Peroxisomal consensus signal |
| CMV (CYTO109-PA-1/CYTO109-VA-1) | pCT-Autophago-GFP | Autophagosome | LC3 |
| CMV (CYTO109R-PA-1/CYTO109R-VA-1) | pCT-Autophago-RFP | Autophagosome | LC3 |
| CMV (CYTO120-PA-1/CYTO120-VA-1) | pCT-CD63-GFP | Exosome/Secretory | CD63 Tetraspanin |
| CMV (CYTO120R-PA-1/CYTO120R-VA-1) | pCT-CD63-RFP | Exosome/Secretory | CD63 Tetraspanin |
| CMV (CYTO122-PA-1/CYTO122-VA-1) | pCT-CD9-GFP | Exosome/Secretory | CD9 Tetraspanin |
| CMV (CYTO123-PA-1/CYTO123-VA-1) | pCT-CD9-RFP | Exosome/Secretory | CD9 Tetraspanin |
| CMV (CYTO124-PA-1/CYTO124-VA-1) | pCT-CD81-GFP | Exosome/Secretory | CD81 Tetraspanin |
| CMV (CYTO125-PA-1/CYTO125-VA-1) | pCT-CD81-RFP | Exosome/Secretory | CD81 Tetraspanin |
| CMV (CYTO110-PA-1/CYTO110-VA-1) MSCV (CYTO110-PB-1/CYTO110-VB-1) | pCT-Actin-GFP | Cytoskeleton | β-actin |
| CMV (CYTO111-PA-1/CYTO111-VA-1) MSCV (CYTO111-PB-1/CYTO111-VB-1) | pCT-Tublin-GFP | Cytoskeleton | α-tublin |
| CMV (CYTO111R-PA-1/CYTO111R-VA-1) | pCT-Tublin-RFP | Cytoskeleton | α-tublin |
| CMV (CYTO112-PA-1/CYTO112-VA-1) | pCT-MAP4-GFP | Microtubule | Microtubule associated protein 4 (MAP4) |
| CMV (CYTO113-PA-1/CYTO113-VA-1) MSCV (CYTO113-PB-1/CYTO113-VB-1) | pCT-H2B-GFP | Nucleus | H2B |
| CMV (CYTO114-PA-1/CYTO114-VA-1) | pCT-Apoptosis-Luc | Caspase 3/7 activation | Cyclic inactive luciferase |
| CMV (CYTO115-PA-1/CYTO115-VA-1) | pCT-GFP-BAX | Cytosol to Mitochondria | BAX |
| CMV (CYTO116-PA-1/CYTO116-VA-1) | pCT- Catenin-GFP | Cytosol to Nucleus/Cell Membrane | β-Catenin activation |
| CMV (CYTO117-PA-1/CYTO117-VA-1) | pCT-CMV-PSD95-GFP | Dendrite Membranes | PSD-95 |
| CMV (CYTO118-PA-1/CYTO118-VA-1) MSCV (CYTO118-PB-1/CYTO118-VB-1) | pCT-Cyto-GFP | Cytosolic Compartment | GFP-Untagged |
| CMV (CYTO119-PA-1/CYTO119-VA-1) | pCT-Cyto-RFP | Cytosolic Compartment | RFP-Untagged |
| MSCV (CYTO121-PB-1/CYTO121-VB-1) | pCT-Cyto-GFpLuc | GFP and Luciferase Control | GFP and Luciferase (Untagged) |
References
How It Works
Supporting Data
Supporting Data
See some of our exosome Cyto-Tracers in action
The following figure and videos are from:
Garcia NA, et al. Glucose Starvation in Cardiomyocytes Enhances Exosome Secretion and Promotes Angiogenesis in Endothelial Cells. PLoS ONE. 2015. 10(9). PMCID: PMC4578916.
Figure 4 from Garcia, et al. Exosome transfer from CMs to ECs. (A) H9c2 transfected with pCT-CD63-GFP. FACS analysis of 90% GFP positive cells. (B) Representative images from confocal time-lapse microscopy of mouse ECs (ACTB-DsRed) (EC; red) co-cultured with H9C2-CD63-GFP cells (green), previously cultured for 24 h in +/-St medium. Exosome transfer from H9C2 CMs to EC can be observed (S1 and S2 Movies, below). White arrows show CD63-GFP structures inside ECs (C) Representative immunostaining of H9C2-CD63-GFP and HUVEC co-cultures; anti-GFP (green) and anti-CD31 (red). The images illustrate GFP fluorescence from CD63-GFP exosomes in CD31-positive cells (red) after 24 h incubation in +/-St medium. White arrows show CD63-GFP structures inside ECs.
Video S1 from Garcia, et al. Exosome transfer from H9C2-CD63-GFP (green) to endothelial DsRed cells (red) under +St conditions.
Video S2 from Garcia, et al. Exosome transfer from H9C2-CD63-GFP (green) to endothelial DsRed cells (red) under -St conditions.
Labeled exosomes. (Top panels) CD63-GFP Cyto-Tracers transfected into a human fibrosarcoma cell line. (Bottom panels) CD9-GFP and CD9-RFP Cyto-Tracers co-transfected into HEK293 cells.